AI-Assisted Clinical Trial Protocol Design Market to Reach USD 5,110.0 million by 2036 Amid Rapid AI-Driven Clinical Trial Digitization
Friday, 15 May 2026 09:50 AM
Company Update
AI-Assisted Clinical Trial Protocol Design Market Driven by Rising Demand for Protocol Optimization, Regulatory-Ready AI Systems, Workflow Automation, and Sponsor-Facing SaaS Platforms to Register 20.8% CAGR Growth Through 2036
NEWARK, DE / ACCESS Newswire / May 15, 2026 / According to the latest market analysis by Future Market Insights (FMI), the AI-assisted clinical trial protocol design market is entering a high-growth expansion phase as pharmaceutical sponsors, CROs, and biotech companies increasingly prioritize AI-enabled systems capable of improving protocol efficiency, reducing operational burden, and accelerating clinical trial timelines.
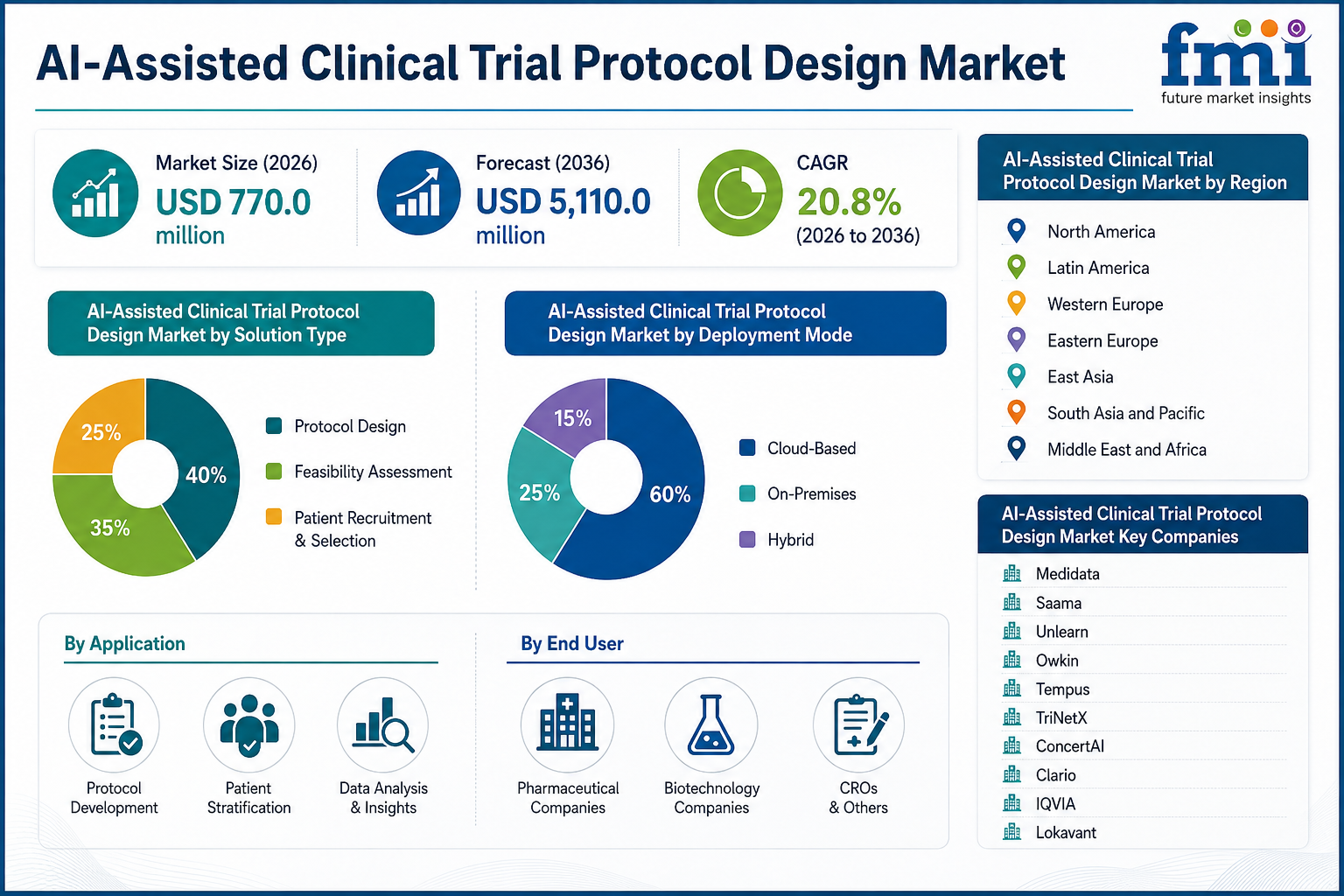
The market, valued at USD 770.0 million in 2026, is projected to reach USD 5,110.0 million by 2036, registering a CAGR of 20.8% during the forecast period. Growth is being supported by increasing adoption of protocol feasibility AI tools, rising demand for sponsor-facing SaaS platforms, and stronger industry focus on operational efficiency, patient recruitment optimization, and regulatory-ready AI deployment across global clinical research ecosystems.
AI-assisted clinical trial protocol design platforms are evolving from exploratory pilot programs into operationally integrated systems supporting trial feasibility, eligibility optimization, patient burden modeling, site selection analytics, and regulatory document automation.
Summary of AI-Assisted Clinical Trial Protocol Design Market
Market Size (2026): USD 770.0 Million
Market Size (2036): USD 5,110.0 Million
Growth Rate: 20.8% CAGR (2026-2036)
Leading Technology Segment: Protocol Feasibility and Optimization Tools (32.0% share)
Leading Trial Phase Segment: Phase II and Phase III Trials (49.0% share)
Leading Buyer Model: Sponsor-Facing SaaS Platforms (52.0% share)
Incremental Opportunity: USD 4,340.0 Million
Key Growth Regions: India, China, United States, United Kingdom, Germany
Key Players: Medidata, Saama, Unlearn, Owkin, Tempus, TriNetX, ConcertAI, Clario, IQVIA, Lokavant
Get detailed market forecasts, competitive benchmarking, and pricing trends:
https://www.futuremarketinsights.com/reports/sample/rep-gb-33074
AI-Driven Operational Efficiency Reshaping Clinical Trial Protocol Development
The AI-assisted clinical trial protocol design market is witnessing strong momentum as pharmaceutical and biotech organizations increasingly seek intelligent systems capable of reducing manual workload, improving protocol quality, and supporting faster decision-making across complex clinical development workflows. Vendors are increasingly focusing on:
Expanding protocol feasibility AI and eligibility optimization capabilities
Developing workflow-integrated sponsor-facing SaaS platforms
Enhancing regulatory-ready AI documentation and traceability systems
Strengthening integration with clinical data management ecosystems
Supporting scalable deployment through managed implementation services
This transition reflects broader healthcare industry demand for operationally practical AI systems that combine measurable workflow value, compliance support, implementation transparency, and repeatable deployment outcomes.
Product and Segment Trends
Market segmentation highlights strong demand for deployable, workflow-integrated AI systems:
Protocol feasibility and optimization tools (32.0%) lead technology demand due to direct operational value and measurable implementation outcomes
Phase II and Phase III trials (49.0%) dominate trial-phase demand because of higher operational complexity and stronger budget allocation
Sponsor-facing SaaS platforms (52.0%) lead buyer adoption through lower implementation friction and recurring workflow integration
Eligibility criteria optimization tools continue gaining momentum as sponsors seek cleaner datasets and improved patient recruitment efficiency
Regulatory document assistants are increasingly supporting audit-ready clinical trial documentation and submission workflows
AI-assisted platforms are increasingly becoming central to clinical trial operational planning, workflow automation, and protocol performance optimization.
Regulatory Readiness, AI Governance, and Workflow Integration Driving Innovation
AI-assisted clinical trial protocol design vendors are prioritizing:
Audit-ready AI systems supporting regulatory credibility assessment frameworks
Scalable SaaS deployment models with repeatable implementation support
Advanced analytics for patient burden modeling and site selection optimization
AI systems integrated with existing clinical operations and enterprise workflows
Service-led deployment and workflow customization capabilities
Innovation is increasingly focused on balancing automation efficiency, integration reliability, compliance transparency, and measurable clinical operations value.
Access the Complete Report in PDF Format:
https://www.futuremarketinsights.com/reports/brochure/rep-gb-33074
Sponsor-Facing SaaS Platforms and Managed Services Driving Value Creation
Technology providers are differentiating through:
Expansion of AI-enabled protocol design and optimization platforms
Strengthening sponsor and CRO implementation support services
Developing scalable workflow integration and monitoring systems
Enhancing data traceability and operational reporting capabilities
Supporting deployment models aligned with regulated clinical environments
Companies capable of combining domain expertise, implementation support, and operationally measurable AI outcomes are gaining competitive advantage across global clinical trial ecosystems.
Supply Chain and Deployment Trends
The market is witnessing increased investment in AI infrastructure and deployment scalability, including:
Expansion of cloud-based sponsor-facing SaaS platforms
Growth in AI workflow integration and managed implementation services
Increased investment in regulatory-compliant data governance systems
Stronger focus on deployment support and post-implementation optimization
Greater emphasis on auditability, workflow traceability, and performance documentation
These strategies are helping suppliers improve customer retention, recurring revenue generation, and enterprise-level deployment scalability.
Regional Outlook and Growth Opportunities
The market demonstrates strong regional development trends:
India (22.6% CAGR): Expanding pharmaceutical R&D activity and growing digital clinical infrastructure supporting rapid adoption
China (21.1% CAGR): Large-scale clinical trial expansion and increasing AI integration accelerating market growth
United States (20.1% CAGR): Mature biotech ecosystem and strong regulatory engagement supporting enterprise deployment
United Kingdom (19.4% CAGR): Increasing AI-driven healthcare innovation and clinical research digitization strengthening adoption
Germany (18.7% CAGR): Strong compliance-focused clinical operations supporting demand for traceable AI systems
The global AI-assisted clinical trial protocol design market remains one of the fastest-growing healthcare technology categories as pharmaceutical sponsors increasingly prioritize operational efficiency, regulatory transparency, and AI-supported protocol optimization worldwide.
Buyer Trends and Enterprise Purchasing Priorities
Key buyer priorities include:
Workflow integration and operational scalability
Regulatory-ready AI systems with auditability
Reduced manual workload and improved protocol efficiency
Measurable deployment outcomes and recurring value creation
Strong implementation support and post-deployment services
Buyers are increasingly prioritizing AI vendors capable of delivering operational reliability, workflow compatibility, and long-term deployment support rather than broad exploratory AI claims.
Competitive Landscape
The AI-assisted clinical trial protocol design market remains fragmented across specialist AI platform providers, workflow integration vendors, and managed service providers focusing on clinical operations optimization.
Leading companies are focusing on:
Expanding AI-enabled clinical trial workflow capabilities
Strengthening sponsor and CRO integration partnerships
Enhancing deployment support and recurring SaaS delivery models
Supporting audit-ready AI documentation and workflow transparency
Investing in scalable AI deployment and operational reporting systems
Key companies include:
Medidata
Saama
Unlearn
Owkin
Tempus
TriNetX
ConcertAI
Clario
IQVIA
Lokavant
Companies capable of combining clinical domain expertise, workflow integration depth, measurable implementation outcomes, and long-term service support are expected to maintain the strongest market positions.
Buy Report: Unlock 360° insights for strategic decision making and investment planning:
https://www.futuremarketinsights.com/checkout/33074
Long-Term Deployment Strategies and Customer Retention
Market participants are increasingly focusing on long-term customer retention through:
Continuous AI workflow optimization and feature expansion
Enhanced sponsor support and deployment customization services
Expansion of regulatory-ready AI governance frameworks
Long-term SaaS subscription and managed service contracts
Workflow monitoring, analytics, and operational reporting enhancements
Vendors delivering integration reliability, operational transparency, measurable workflow improvements, and scalable deployment support are expected to achieve stronger recurring revenue and long-term market positioning.
Related Reports
Clinical Trials Market - https://www.futuremarketinsights.com/reports/clinical-trials-market
Clinical Trial Data Management Service Market - https://www.futuremarketinsights.com/reports/clinical-trial-data-management-service-market
Clinical Trial Biorepository & Archiving Solution Market - https://www.futuremarketinsights.com/reports/clinical-trial-biorepository-and-archiving-solutions-market
Allergy Clinical Trial Services Market - https://www.futuremarketinsights.com/reports/allergy-clinical-trial-services-market
AI-based Clinical Trials Solution Provider Market - https://www.futuremarketinsights.com/reports/ai-based-clinical-trials-solution-provider-market
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
For Press & Corporate Inquiries
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - [email protected]
For Media - [email protected]
For web - https://www.futuremarketinsights.com/
For Web - https://www.factmr.com/
SOURCE: Future Market Insights, Inc.

